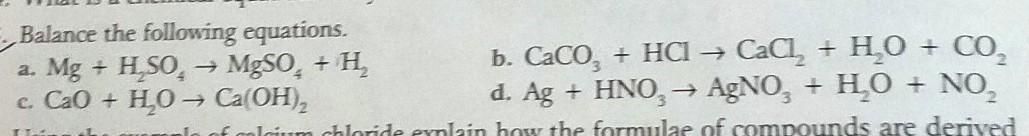Answers
Answer:
A) Mg + H2SO4 → MgSO4 + H2
B) CaCO3 + 2HCl → CaCl2 + H2O + CO2
C) CaO + H2O → Ca(OH)2
D) 3Ag + 4HNO3 → 3AgNO3 + H2O + NO2
Explanation:
A) Mg + H2SO4 → MgSO4 + H2
The equation is already balanced because the number of each element in the left hand side is already equal to that in the right hand side.
B) CaCO3 + 2HCl → CaCl2 + H2O + CO2
This equation is balanced by putting 2 in front of HCl on the left hand side to balance the elements on the right hand side.
C) CaO + H2O → Ca(OH)2
Thus equation is already balanced because the number of each element in the left hand side is already equal to that in the right hand side.
D) 3Ag + 4HNO3 → 3AgNO3 + H2O + NO2
This equation is balanced by putting 3 in front of Ag and 4 in front of HNO3 on the left hand side while we put 3 in front of AgNO3 on the right side to balance the elements on the right hand side.
Related Questions
Two samples of hot unknown metals of equal temperatures are dropped in separate containers of water at 30 degrees Celsius. The final temperature of sample A is 33.5 degrees, and the final temperature of sample B is 31.8 degrees. Which metal sample had the highest specific heat?
Answers
Answer:
Sample B has the highest specific heat
Explanation:
Specific heat of a material is defined as the heat necessary to increase in the temperature of 1g of material in 1°C.
The material with the highest specific heat requires more energy to increase its temperature.
In the problem, sample A and B are under the same amount of energy but sample A increases more its temperature. That means sample A has the lowest specific heat and:
Sample B has the highest specific heatPlease help is for my final An example of a chemical change is-
evaporation of seawater
о
sublimation of dry ice (CO2)
melting of aluminum
burning wood reducing it to ashes
Answers
Explanation:
Souring of milk
Baking of cake
Calculate the number of moles of 2.00g of K2SO4
Answers
Which of the following can be used to measure an object's
speed?
Answers
Answer:
distance traveled divided by the time it traveled
Explanation
this is just an equation for how to find speed. you can also just use like a meterstick and a stopwatch
How much of a 10 M solution is needed to make 1 liter of a 1M solution?
a. 1ml
b. 1000ml
c. 10ml
d. 100ml
Answers
i dont know I hope someone knows soon take care
According to molar concentration, 100 ml of 10 M solution is needed to make 1 liter of a 1 M solution.
What is molar concentration?Molar concentration is defined as a measure by which concentration of chemical substances present in a solution are determined. It is defined in particular reference to solute concentration in a solution . Most commonly used unit for molar concentration is moles/liter.
The molar concentration depends on change in volume of the solution which is mainly due to thermal expansion. Molar concentration is calculated by the formula, molar concentration=mass/ molar mass ×1/volume of solution in liters.
In terms of moles, it's formula is given as molar concentration= number of moles /volume of solution in liters.In case of two solutions, molar concentration is found out by, M₁V₁=M₂V₂.
On substitution in above formula, V₁=1×1/10=0.1 L=100 ml.
Thus, 100 ml of a 10 M solution is needed to make 1 liter of a 1 M solution.
Learn more about molar concentration,here:
https://brainly.com/question/21841645
#SPJ2
Identify the two acids and the two bases out of the 5 chemicals below: NaOH/ HCI /H2SO4/ Ca(OH)2 /NaBr *
Answers
Answer: NaOH and Ca(OH)2 are bases. HCl and H2SO4 are acids
Explanation:
Zinc reacts with Copper (II) chloride to produce Copper and Zinc chloride. -write chemical formula
Answers
Answer:
here's the answer hope it helps
Mention any two uses of oxygen gas.
Answers
Answer:
steel, plastics
Explanation:Hope it helps!
Pls help on test (no old answers OR files)
During a combustion reaction, 9.00 grams of oxygen reacted with 3.00 grams of CH4.
What is the amount of the leftover reactant?
0.74 grams of methane
0.89 grams of methane
1.22 grams of oxygen
1.45 grams of oxygen
Answers
What is the molar mass of H2S?
0 518
65.15 g
17 g
78.16 g
34.1g
Answers
S - 32.1
(1*2) + 32 = 2 + 32.1 = 34.1
34.1 g is the answer
The air we breathe contains different individual gases (mostly nitrogen and oxygen). Which of the following correctly describes the air we breathe?
A. homogeneous mixture
B. compound
C. heterogeneous mixture
D. element
Answers
Which of the following represents alpha decay?
Answers
Answer: b
Explanation: alpha decay or particle consist of two protons and two neutrons hence it is a helium nucleus.
An alpha particle [tex]\binom {4} {2}[/tex]He is produced in alpha decay reactions, while the parent nucleus' atomic number and mass number drop by 2 and 4 correspondingly.
Alpha decay:Alpha decay is a nuclear process whereby an unstable nuclear transforms into a different element by ejecting particles composed of two protons and two neutrons. The alpha particle also referred to as a helium nucleus, is the expelled unit of matter. Alpha particles have large masses and electrostatic attraction.An atomic number decreases by 2 and the atomic weight drops by 4 when a radioactive nucleus disintegrates by emitting [tex]\bold{\alpha}[/tex] particle.Therefore, the final answer is "Option B".
Find out more about the alpha decay here:
brainly.com/question/14081292
Determine the grams of water produced when 250 grams of barium hydroxide react
with 125 grams of HCI. Refer to the balanced equation below.
Ba(OH)2 (aq) + 2HCl (aq) --> 2H2O (l) + BaCl2 (aq) (balanced)
Answers
Answer:
Explanation:
Ba(OH)₂ (aq) + 2HCl (aq) --> 2H₂O (l) + BaCl₂ (aq)
171 g 2 x 36.5 g 2 x 18 g
73 g of HCl reacts with 171 g of Ba(OH)₂
125 g HCl reacts with 171 x 125 / 73 g of Ba(OH)₂
= 292.8 g of of Ba(OH)₂
Ba(OH)₂ available is 250 g , so it is the limiting reagent .
171 g of Ba(OH)₂ forms 36 g of water
250 g of Ba(OH)₂ forms 36 x 250 / 171 g of water
= 52.63 g of water.
Copper (II) reacts with silver nitrate in a single displacement reaction. How many grams of copper are needed to react with 510 grams of silver nitrate?
Answers
The answer is 95.39 g Cu
if (H+] = 10^-2 M
what will be pH = ?
Answers
pH = -log(10^-2)
pH = 2
balance CO2+h2=C6H12O6+O2
Answers
Answer:
C 6 H 12 O 6 + 6 O 2 --> 6 CO 2 + 6 H 2 O + ATP is the complete balanced chemical formula for cellular respiration.
Why are shadows shorter around noon?
Make sure you give a full, detailed answer, not just one sentence.
Answers
Answer: Answer is in the Explanation.
Explanation: At noon the sun is directly above in the sky, the sun's rays are falling on the body making the shadow smaller. While in the evenings and mornings the sun is at a inclined position, so the sun's rays are falling at a inclined position making the body's shadow larger.
I looked it up on the internet and reworded it as good as I could.
Explain what will happen to the organisms that is affected by human activities.
Answers
Answer:
Human activity is by far the biggest cause of habitat loss.
Humans are now responsible for causing changes in the environment that hurt animals and plant species. We take up more space on Earth for our homes and cities. We pollute habitats. ... Human activity often changes or destroys the habitats that plants and animals need to survive.
Explanation:
Human activities are the various actions for recreation, living, or necessity done by people. For instance it includes leisure, entertainment, industry, recreation, war, and exercise.
Over time, destruction of such habitats leads to reduced biodiversity, weakening the Earth's ecosystems, and ultimately posing a major threat to human life. While, significant tracts of habitat have been lost, and along with them many species of plant and animal, steps can be taken to slow and even reverse the process.
One of the most significant ways in which humans have impacted on the lives of other species is by causing climate change. Many animals, including birds and frogs, now breed much earlier in spring in Britain than they did 20 years ago. Whether or not this is having a harmful effect on them has yet to be determined.
Helppppppppp pweaseeeee
Answers
Answer:
Explanation:
b
What is the center of the atom called?
Group of answer choices
Electron
Neutron
Nucleus
Proton
Answers
Answer:
nucleus
Explanation:
thats 100% the answer
How many ounces is 68 g?
O A. 0.42 oz
O B. 28.3 oz
O C. 4.8 oz
O D. 2.4 oz
Answers
Answer:
D
Explanation:
How much energy does a copper sample absorb as heat if its specific heat is 0.384 J/g.°C, its mass is 8.00 g, and it is
heated from 10.0°C to 40.0°C?
Answers
Answer:
.0016
Explanation:
For specific heat problems you use the formula :
Q = mcΔT
They've given you the m, c and delta T, so you can plug in those values. (for the temperature change subtract 10 from 40 to see that it grew 30 degrees)
You're looking for Q so leave that variable in the equation. Then solve.
.Q = (8.00)(.384)(30)
Q = 92.16
92.16 J energy will be absorbed by a copper sample.
Given:
Specific heat capacity, C= 0.384 J/g °C
Temperature, T₁ = 10.0°C and T₂= 40.0°C
Mass, m=8.00 g
We know that,
The specific heat capacity is defined as the quantity of heat (J) absorbed per unit mass (g) of the material when its temperature increases or 1 °C, and its unit is J/g °C.
Heat energy in terms of specific heat energy can be calculated by using this formula:
Q= m. C. ΔT
∵ΔT= change in temperature,
ΔT=40.0-10.0°C=30°C
Now substituting the values in the above formula.
[tex]Q= 8.0*0.384*30=92.16J[/tex]
Hence, 92.16 J energy will be absorbed by a copper sample.
Learn more:
brainly.com/question/22991121
List ways on how to lessen human impacts in the environment
Answers
Answer:
Explanation:
Completely turn off equipment like televisions and stereos when you're not using them.
Choose energy-efficient appliances and light bulbs.
Save water.
Lower your shades or close your curtains on hot days, to keep the house cool and reduce the use of electric fans or air-conditioning.
Let clothes dry naturally.
Keep lids on pans when cooking to conserve energy.
Use rechargeable batteries.
Don’t use “throw away” products like paper plates and napkins, or plastic knives, forks and cups.
Send electronic greetings over email, instead of paper cards.
subtracting atomic number from atomic mass will give the number of
Answers
Answer:
:)
Explanation:
If we subtract the atomic number from the atomic mass: atomic mass - atomic number = number of protons + number of neutrons - number of protons. Thus we get the number of neutrons present in an atom when we subtract the atomic number from the atomic mass.
I need help with this…and can you so the work if you can!!
Answers
Answer:
88 grams CO2
Explanation:
Convert from grams to moles to mole ratio to grams again
32 g CH4 * 1 mol CH4/16 g CH4 * 1 mol CO2/1 mol CH4 * 44 grams CO2 = 88 grams CO2
Lemme know if u need more explanation!
How are radar and an approaching siren different? How are they similar?
A. Radar uses sound waves, and the siren uses radio, but both exhibit
the Doppler effect
B. Radar uses radio, and the siren uses sound waves, but both exhibit
the Doppler effect
C. The Doppler effect can be observed only with radar, but both
devices use sound waves.
D. The Doppler effect can be observed only in the approaching siren,
but both devices use radio.
Answers
Answer:
b
Explanation:
Radar uses radio, and the siren uses sound waves, but both exhibit the Doppler effect. Therefore, option (C) is correct.
What is Doppler's effect?Radar operates by emitting radio waves and measuring the reflected waves to detect objects and determine their velocity. On the other hand, an approaching siren emits sound waves that propagate through the air.
Both radar and an approaching siren exhibit the Doppler effect. The Doppler effect is the change in frequency or pitch of waves (sound or electromagnetic) when the source of the waves and the observer are in relative motion. In the case of radar, the Doppler effect is used to measure the velocity of objects based on the frequency shift of the reflected radio waves. Similarly, the Doppler effect is observed with an approaching siren as the sound waves compress and the frequency appears higher as the source approaches the listener.
Therefore, both radar and an approaching siren differ in the type of waves they use (radio waves vs. sound waves), but they share the characteristic of exhibiting the Doppler effect.
Learn more about Doppler effect. here:
https://brainly.com/question/28106478
#SPJ5
Which expression represents the pH of a solution?
Answers
Answer:
pH = 14 - pOH
pH = -log[H+]
pH = pKa + log [A-]/[HA]
Explanation:
The equation used depends on what is given in the equation.
The expression that represent the pH of the solution is
pH = 14 - pOH
pH = -log[H+]
pH = pKa + log [A-]/[HA]
What is the pH of the solution?It is the quantitative measure related to the acidity or the other liquid solutions. It should be range between the 1 and 10^-14. here the numbers be like from 0 to 14.
Hence, we can say that The expression that represent the pH of the solution is
pH = 14 - pOH
pH = -log[H+]
pH = pKa + log [A-]/[HA]
Learn more about pH here: https://brainly.com/question/12052751
Helpppppp ASAP I will give correct answer brainliest 
Answers
Answer: capillaries are not used to exchange substances between the body cells
Explanation:
Arteries carry blood away from the heart. Veins carry blood to the heart except the hepatic portal vein. Capillaries are used to exchange substances between body cells.
C
ot 헤
C.
Show that the results of these dulys
Calculate the oxidation state of Manganese in the following
i. KMnO4 ii. Mno4-
Answers
i) KMnO4
lets calculate using x method
1+x+ 4(-2) = 0
1+x-8 =0
x= 7
ii)MnO4‐
x+4(-2) = -1
x-8 = -1
x = 7
what are the three method used to treat water ?
Answers
Answer:
The three methods used to treat water are
Filtration SedimentationDistillationExplanation:
These methods used include physical processes, biological processes such as slow sand filters or biologically active carbon; chemical processes such as flocculation and chlorination; and the use of electromagnetic radiation such as ultraviolet light.