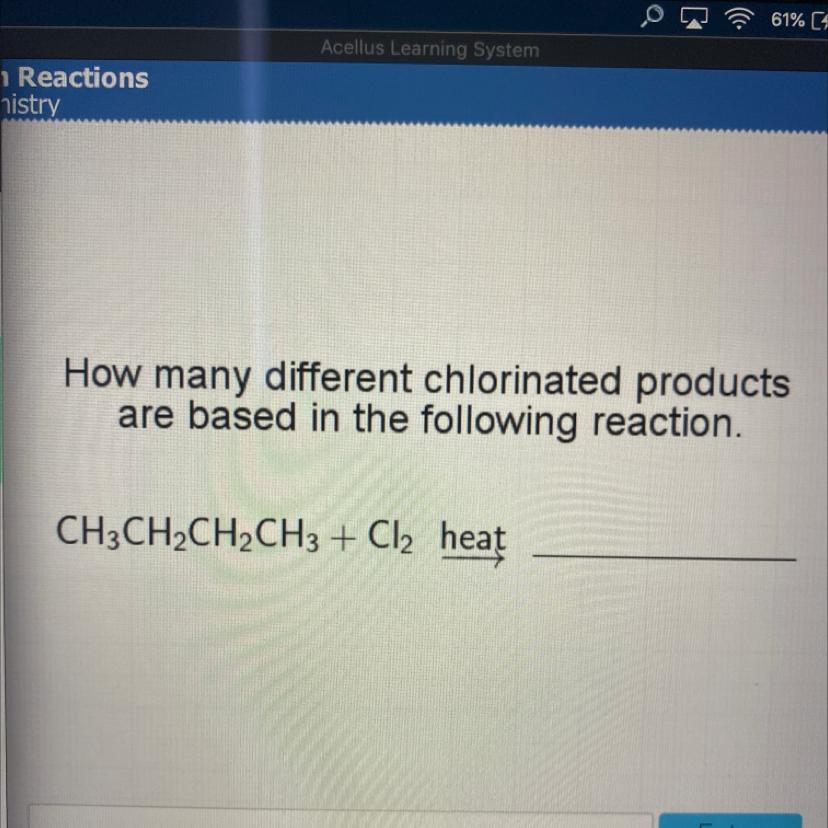
Answers
Answer: if I’m not mistaken it would be 8
Explanation:
Answer:
2
Explanation:
:)
Related Questions
How many hydrogen atoms can be bonded to one carbon atom?
O A. 3
O B. 1
O C. 2.
O D. 4
Answers
Answer:
O D. 4
Explanation:
A carbon atom is an element that can be found in the atomic number 6th of the periodic table. It electronic configuration is 1s² 2s² 2p². It is a tetravalent and non metallic compound. In its outermost structure, Carbon needs four additional valence electrons to complete its octet structure. That is why hydrogen with only one electron can be bonded to one carbon atom in four different places making the carbon element obtain a tetrahedral shape.
to find the mass in g of 3 moles Al .
Answers
Answer:
81 gm
Explanation:
The mass of 1 mole is 27 gSo, Mass of 3 moles of Aluminium would be 3 × 27 = 81 gm
Thus mass of 3 Moles of Aluminium is 81 gm.
Calculate the value of ΔG° at 25°C for the formation of POCl3 from its constituent elements,
P2 (g) + O2 (g) + 3Cl2 (g) --> 2POCl3 (g)
A. -606.2 kJ/molrxn
B. +1108.7 kJ/molrxn
C. -1108.7 kJ/molrxn
D. +606.2 kJ/molrxn
Answers
The correct answer is
C. -1108.7 kJ/mol rxn
From the table values, the standard Gibbs free energy change for the formation of POCl₃ is -1108.7 kJ/mol
What is Gibbs free energy ?We can calculate ΔG° for the reaction using the following formula:
ΔG° = ΣnΔG°f(products) - ΣnΔG°f(reactants)
where ΔG°f is the standard Gibbs free energy of formation for each compound and n is the stoichiometric coefficient of each compound in the balanced chemical equation.
We can look up the values of ΔG°f for each compound in tables of thermodynamic data. At standard conditions (25°C and 1 atm pressure), the values are:
ΔG°f (POCl₃) = -558.9 kJ/mol
ΔG°f (P₂) = 0 kJ/mol
ΔG°f (O₂) = 0 kJ/mol
ΔG°f (Cl₂) = 0 kJ/mol
Plugging these values into the formula, we get:
ΔG° = (2 mol)(-558.9 kJ/mol) - (1 mol)(0 kJ/mol) - (1 mol)(0 kJ/mol) - (3 mol)(0 kJ/mol)
ΔG° = -1117.8 kJ/mol
Therefore, the correct option is C. -1108.7 kJ/molrxn. Note that this answer differs slightly from the calculated value because the given choices are rounded to one decimal place.
Find more on Gibbs energy:
https://brainly.com/question/29753417
#SPJ2
An electric kettle uses electrical energy to boil water. Energy from the electricity is transferred to the water, heating it up. An electric ice maker also uses electrical energy, but it freezes water to form ice. Since energy can’t be created or destroyed, and water loses potential energy when it freezes to form ice, what happens to the energy put into the ice maker and the energy released by the water?
Answers
Answer:
Since energy can’t be created or destroyed, and water loses potential energy when it freezes to form ice. The energy remains electrical but then changes to kinetic when Enough energy is released.
Explanation:
The excess electrical energy gained by water during ice forming process is removed as potential energy to maintain law of conservation of energy.
According to the principle of conservation of energy, energy can neither be created nor destroyed but can transformed from one form to energy.
The electrical energy used by the ice maker to freeze water works on the same principle of conservation of energy.
The electrical energy absorbed the water is used to lower the temperature of the water in order to form ice by removing heat from the water in form of potential energy. This happens to maintain the principle of conservation of energy.Thus, the excess electrical energy gained by water during ice forming process is removed as potential energy to maintain law of conservation of energy.
Learn more about conservation of energy here: https://brainly.com/question/166559
a transverse wave moves a medium at an abtuse angle to the wave , true or false ?
Answers
Answer:
True! A transverse wave does move a medium at an obtuse angle to the wave!!
Answer:
False
Explanation:
A student is titrating 50ml of 2.5M HI with 1.23M NaOH. How many milliliters of base are added to reach the
equivalence point?
Answers
Answer:
101.6ml
Explanation:
Using the formula;
CaVa = CbVb
Where;
Ca = concentration of acid (M)
Cb = concentration of base (M)
Va = volume of acid (ml)
Vb = volume of base (ml)
Based on the information as follows:
Ca = 2.5M
Cb = 1.23M
Va = 50ml
Vb = ?
Using CaVa = CbVb
2.5 × 50 = 1.23 × Vb
125 = 1.23Vb
Vb = 125/1.23
Vb = 101.6ml
does the moon really change shape? why
Answers
what is chemical name of ghee
Answers
Answer. Chemical formula of ghee: (C17H33 Coo)3 C3H5 • Chemical Reaction: (C17H31Coo)3 C3H5 + 3H2 (C17H33 Coo)3 C3H5
If only 0.186 g of Ca(OH)2 dissolves in enough water to give 0.230 L of aqueous solution at a given temperature, what is the Ksp value for calcium hydroxide at this temperature?
Answers
Answer:
[tex]Ksp=5.20x10^{-6}[/tex]
Explanation:
Hello there!
In this case, according to the solubility equilibrium of calcium hydroxide:
[tex]Ca(OH)_2\rightleftharpoons Ca^{2+}+2OH^-[/tex]
Whereas the equilibrium expression is:
[tex]Ksp=[Ca^{2+}][OH^-]^2[/tex]
It is firstly necessary to calculate the molar solubility given the grams and volume of the dissolved solute:
[tex]s=\frac{0.186g/(74.09g/mol)}{0.230L}=0.0109M[/tex]
Now, according to the Ksp expression, we plug in s as the solubility to obtain:
[tex]Ksp=(s)(2s)^2\\\\Ksp=(0.109)(2*0.0109)^2\\\\Ksp=5.20x10^{-6}[/tex]
Regards!
According to the solution equilibrium,
[tex]Ca(OH)_2 \rightleftharpoons Ca^{2+}+2OH^-[/tex]Now,
The molar solubility will be:
→ [tex]s = \frac{\frac{0.186}{74.09} }{0.230}[/tex]
[tex]= 0.0109 \ M[/tex]
hence,
The Ksp value will be:
→ [tex]Ksp = (s)(2s)^2[/tex]
By substituting the values,
[tex]= (0.109)(2\times 0.0109)^2[/tex]
[tex]= 5.20\times 10^{-6}[/tex]
Thus the above approach is right.
Learn more about temperature here:
https://brainly.com/question/23773843
The total pressure in a tank is filled with a mixture of gases : Oxygen, Helium, and Argon is 15.3 atm. The P(He) = 3.2 atm, P(O) = 7.4 atm, what is the P(Ar) in the tank?
Answers
Answer:
4.7 atm
Explanation:
From the question given above, the following data were obtained:
Total pressure (P) = 15.3 atm
Pressure of Helium, P(He) = 3.2 atm
Pressure of Oxygen, P(O) = 7.4 atm
Pressure of Argon, P(Ar) =?
The pressure of Argon, P(Ar) in the tank can be obtained as illustrated below:
P = P(He) + P(O) + P(Ar)
15.3 = 3.2 + 7.4 + P(Ar)
15.3 = 10.6 + P(Ar)
Collect like terms
15.3 – 10.6 = P(Ar)
4.7 = P(Ar)
P(Ar) = 4.7 atm
Therefore, the pressure of Argon, P(Ar) in the tank is 4.7 atm
Using the Lewis dot structures of magnesium and oxygen, predict the ionic formula
Answers
Answer:
(edit: nvm I figured it out, here is the answer)
Explanation:
Ions exists bound together in ionic formulae by electrostatic attraction then the ionic formula exists MgO.
What is meant by Lewis dot structures?The diagrams known as Lewis structures, also referred to as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures, depict the interactions between the atoms in a molecule as well as any lone pairs of electrons that may be present.
A chemical compound comprehended as an ionic compound in chemistry exists one that contains ions bound together by the electrostatic forces named as ionic bonding. The molecule exists often neutral despite containing both positively and negatively charged ions, or cations and anions. Ions exists bound together in ionic formulae by electrostatic attraction.
Magnesium and oxygen:
Cation [tex]$=\mathrm{Mg}^{2+}$[/tex]
Anion [tex]$=\mathrm{O}^{2-}$[/tex]
Net charge = + 2 - 2 = 0
Formula = MgO
Therefore, the ionic formula exists MgO.
To learn more about ionic compound refer to:
https://brainly.com/question/2687188
#SPJ2
what are the compounds that cause acid rain?
Answers
Answer:
Explanation:
Its a chemical reaction that begins when compounds like sulfur dioxide and nitrogen oxides are released into the air. These substances rise high into the atmosphere in which they mix and react with water, oxygen, and other chemicals to form more acidic pollutants.
Answer:
Explanation:
Acid rain is caused by a chemical reaction that begins when compounds like sulfur dioxide and nitrogen oxides are released into the air. These substances can rise very high into the atmosphere, where they mix and react with water, oxygen, and other chemicals to form more acidic pollutants, known as acid rain.
PLEASE HELP, DUE AT 12:00
Answers
pls help i need help help
Answers
Answer:
and is capillaries
Explanation:
hope it helps
Which of the following indicates that a chemical equation is balanced?
a. The numbers of atoms of each element are the same on both sides of the equation.
b. All of the coefficients are the same.
C. The numbers of molecules on each side of the equation are equal.
d. The sums of the coefficients on each side of the equation are equal.
Answers
Match the solutions to the descriptions of the freezing points.a. One mole of the ionic compound Na3PO4 dissolved in 1000 g H2O b. One mole of the ionic compound CuSO4 dissolved in 1000 g H2O c. One mole of the nonelectrolyte C6H12O6 dissolved in 1000 g H2O1. Highest freezing point 2. Intermediate freezing point 3. Lowest freezing point
Answers
Explanation:
Depression in Freezing point
= Kf × i × m
where m is molality , i is Van't Hoff factor, m = molality
Since molality and Kf remain the same
depression in freezing point is proportional to i
i= 2 for CuSO4 ( CuSO4----------> Cu+2 + SO4-2
i=1 for C2h6O
i= 3 for MgCl2 ( MgCl2--------> Mg+2+ 2Cl-)
So the freezing point depression is highest for MgCl2 and lowest for C2H6O
so freezing point of the solution = freezing point of pure solvent- freezing point depression
since MgCl2 has got highest freezing point depression it will have loweest freezing point and C2H6O will have highest freezing point
Calculate the number of molecules in 14.3 mol H2O.
Answers
Answer:
I don't get it
Explanation:
please explain more about question
If a particular ore contains 56.3 % % calcium phosphate, what minimum mass of the ore must be processed to obtain 1.00 kg k g of phosphorus?
Answers
Answer:
34.44 kg
Explanation:
First we convert 1.00 kg of phosphorus (P) into moles, using its molar mass:
1.00 kg ÷ 32 kg/kmol = 0.03125 kmol PThen we convert 0.03125 kmoles of P into kmoles of Ca₃(PO₄)₂:
0.03125 kmol P * [tex]\frac{2kmolCa_3(PO_4)_2}{2kmolP}[/tex] = 0.0625 kmol Ca₃(PO₄)₂Now we calculate the mass of 0.0625 kmoles of Ca₃(PO₄)₂:
0.0625 kmol Ca₃(PO₄)₂ * 310.18 kg/kmol = 19.39 kgFinally we calculate the required mass of the ore, using the definition of content percentage:
% content = Mass of calcium phosphate / mass of ore * 100 %56.3 % = 19.39 kg / mass of ore * 100%Mass of Ore = 34.44 kgTRUE or FALSE: The only way to see a recessive trait is to inherit 2 recessive
alleles.
Answers
Answer:
false it's more than two
7
State two signification of melting?
Answers
what is a major crack in the earth's surface?
Answers
Answer:
A crack in earth's crust is called a fault also known as a "fracture"
Faults are also referred to as fractures.
Scientist use models of the solar system to
1 avoid the use of incorrect data
2 prevent duplication of their ideals
3 help explain their ideas
4 make theories non-testable
Answers
Question 2 (1 point)
An object's gravitational force depends primarily on the object's
a
density
mass
b
oc
momentum
Answers
Answer: An object's gravitational force depends primarily on the object's mass.
Explanation:
According to the Universal law of gravitation, every object whether is it is having large r small mass tends to exert a force on every other object, therefore this force is known as gravitational force.
Formula to calculate gravitational force is as follows.
[tex]F = \frac{G \times m_{1} \times m_{2}}{r^{2}}[/tex]
where,
F = Gravitational force
G = Gravitational constant = [tex]6.674 \times 10^{-11} m^{3}/kg s^{2}[/tex]
[tex]m_{1}[/tex] = mass of object 1
[tex]m_{2}[/tex] = mass of object 2
r = distance between the centers of the objects
Therefore, it means that gravitational force of an object primarily depends on an object's mass.
Thus, we can conclude that an object's gravitational force depends primarily on the object's mass.
2. Acceleration is______
proportional to mass,
Answers
Answer:
Inversely
Explanation:
The acceleration of an object as produced by a net force is directly proportional to the magnitude of the net force, in the same direction as the net force, and inversely proportional to the mass of the object.
When of glycine are dissolved in of a certain mystery liquid , the freezing point of the solution is lower than the freezing point of pure . On the other hand, when of ammonium chloride are dissolved in the same mass of , the freezing point of the solution is lower than the freezing point of pure . Calculate the van't Hoff factor for ammonium chloride in .
Answers
The given question is incomplete. The complete question is:
When 282. g of glycine (C2H5NO2) are dissolved in 950. g of a certain mystery liquid X, the freezing point of the solution is 8.2C lower than the freezing point of pure X. On the other hand, when 282. g of ammonium chloride are dissolved in the same mass of X, the freezing point of the solution is 20.0C lower than the freezing point of pure X. Calculate the van't Hoff factor for ammonium chloride in X.
Answer: the van't Hoff factor for ammonium chloride is 1.74
Explanation:
Depression in freezing point is given by:
[tex]\Delta T_f=i\times K_f\times m[/tex]
[tex]\Delta T_f=T_f^0-T_f=8.2^0C[/tex] = Depression in freezing point
[tex]K_f[/tex] = freezing point constant = ?
i = 1 ( for non electrolyte)
m= molality
[tex]8.2^0C=1\times K_f\times \frac{\text{mass of solute}}{\text{molar mass of solute}\times \text{weight of solvent in kg}}[/tex]
Weight of solvent (X)= 950 g = 0.95 kg
Molar mass of solute (glycine) = 75.07 g/mol
Mass of solute (glycine) = 282 g
[tex]8.2^0C=1\times K_f\times \frac{282g}{75.07g/mol\times 0.95kg}[/tex]
[tex]K_f=2.07[/tex]
ii) [tex]20.0^0C=i\times \times \frac{\text{mass of solute}}{\text{molar mass of solute}\times \text{weight of solvent in kg}}[/tex]
Weight of solvent (X)= 950 g = 0.95 kg
Molar mass of solute (ammonium chloride) = 53.49 g/mol
Mass of solute (ammonium chloride) = 282 g
[tex]20.0^0C=i\times 2.07\times \frac{282g}{53.49g/mol\times 0.95kg}[/tex]
[tex]i=1.74[/tex]
Thus the van't Hoff factor for ammonium chloride is 1.74
According to the following reaction, how many grams of
hydrobromic acid are needed to form 24.7 grams of bromine?
hydrobromic acid (aq) —>hydrogen (g) + bromine (1)
…grams hydrobromic acid
Answers
Answer:
25.1 g
Explanation:
Step 1: Write the balanced decomposition reaction
2 HBr(aq) ⇒ H₂(g) + Br₂(l)
Step 2: Calculate the moles corresponding to 24.7 g of Br₂
The molar mass of Br₂ is 159.81 g/mol.
24.7 g × 1 mol/159.81 g = 0.155 mol
Step 3: Calculate the moles of HBr needed to produce 0.155 moles of Br₂
The molar ratio of HBr to Br₂ is 2:1. The moles of HBr needed are 2/1 × 0.155 mol = 0.310 mol.
Step 4: Calculate the mass corresponding to 0.310 moles of HBr
The molar mass of HBr is 80.91 g/mol.
0.310 mol × 80.91 g/mol = 25.1 g
In a rush, a student only measured the mass of the empty beaker once before beginning
the experiment and one more time after the entire experiment was finished. The mass of
the empty beaker was found to be 0.123g less at the end of the experiment. What
happened?
Answers
Answer:
The student measured the cup and got an incorrect answer
Explanation:
because he was in a rush.
Kantahan moko sa lobby ml tayo babae 11 years old mayhem folow me 908723751 Mobile Legends Bang bang
Answers
Answer:
hi which language is this???
I can't understand
Answer:
Filipino hello pari omsim
match the molecular shapes to the correct Lewis structures
trigonal pyramid
trigonal planar
bent
tetrahedral
linear
Answers
Answer:
Match up correct answers:
1) trigonal planar
2) tetrahedral
3) trigonal pyramid
4) linear
Explanation:
(correct answers on portal and edmentum users)
Hope I was helpful, have a great day and good luck :)
AlH₃ - trigonal planar
CH₂F₂ - tetrahedral
PH₃ - trigonal pyramidal
O₃ - bent
What is a Lewis structure?The valence shell electrons in a molecule are depicted in an extremely simplified manner by a Lewis Structure. It is used to demonstrate how the electrons in a molecule are positioned around particular atoms. Electrons are shown as dots or, in the case of a bond, as a line connecting the two atoms.
How do you determine the molecular shape?Depict the Lewis Structure.Determine if the electron groups are in bond pairs or are lone pairs by counting the number of electron groups. Keep in mind that lone pairs and bonds are both included in electron groups!The electron-group geometry is named. (Specify if it is octahedral, tetrahedral, trigonal-bipyramidal, or linear.)The molecular geometry is determined by examining the locations of the surrounding atomic nuclei. (Count the number of lone pairs)Learn more about molecular geometry here:
https://brainly.com/question/7558603
#SPJ2
Magnesium nitride, Mg3N, undergoes a thermo decomposition reaction to form magnesium metal and nitrogen gas.
a. True
b. False
Answers
Answer:
a. True
Explanation:
Magnesium nitride, Mg₃N, is a binary salt. Upon heating, it decomposes into magnesium metal and nitrogen gas. The unbalanced equation is:
Mg₃N(s) ⇒ Mg(s) + N₂(g)
We can balance it with the trial and error method. First, we will balance N atoms by multiplying Mg₃N by 2.
2 Mg₃N(s) ⇒ Mg(s) + N₂(g)
Then, we get the balanced equation by multiplying Mg by 6.
2 Mg₃N(s) ⇒ 6 Mg(s) + N₂(g)