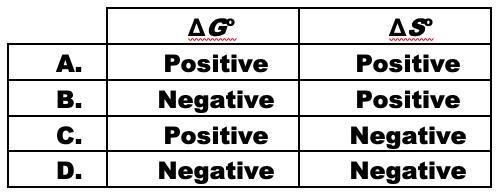
Answers
B.
Delta G is negative and Delta S is positive.
Related Questions
If 0.60 L of a solution contains 6.6 g of NaBr, what is its molar concentration?
Answers
Answer:
Molarity = moles ÷ liters
to get moles of NaBr divide grams of NaBr by its molar mass (mass of Na + mass of Bromine)
Na = 22.989769
Br = 79.904
molar mass of NaBr = 102.893769
6.6g ÷ 102.893769 = 0.064143826 moles of NaBr
0.064143826 moles ÷ 0.60 liters = 0.1069 molar concentration or 11 %
what cause a mass defect?
○A. The mass of a nucleus is larger than it should be.
○B. The mass of a nucleus cannot be accurately measured.
○C. Mass is converted to the energy binding a nucleus together.
○D. Mass is lost when a particle is released in a reaction.
Answers
Answer: Mass is converted to the energy binding a nucleus together.
Explanation: a p e x
Mass is converted to the energy binding a nucleus together cause a mass defect.Hence , Option (C) is correct.
What is Mass defect ?
The actual atomic mass is less than the predicted mass calculated by adding the masses of nucleons.
This additional mass is accounted for by binding energy that is released when a nucleus is formed.
When a nucleus is formed, some of the mass is converted to energy and this results in the mass defect.
Therefore, Mass is converted to the energy binding a nucleus together cause a mass defect. Hence , Option (C) is correct.
Learn more about mass defect here ;
https://brainly.com/question/11624098
#SPJ2
Which atoms are most likely to form + 1 ion and -2 ion respectively?
a. Sr and S
b. Kand o
C. Fe and N
d. Ba and F
Answers
How many liters of a 0.689 M solution of NaCl would contain 75 g of NaCl?
Answers
Answer:
E) the K+ ions are attracted to the partially positive hydrogen atoms of the water molecule. D) the K+ ions are attracted to the partially negative oxygen atoms of the water molecule. Water is a polar solvent and hexane (C6H14) is a nonpolar solvent. Which of the following correctly describes the solubility of the solute? A) weak electrolyte.
Explanation:
A gas has a solubility of 2.45 g/L at a pressure of 0.750 atm. What pressure wold be required to produce an aqueous solution containing 6.25 g/L of this gas at constant temperature
Answers
Answer:
1.91 atm
Explanation:
Step 1: Calculate Henry's constant (k)
A gas has a solubility (C) of 2.45 g/L at a pressure (P) of 0.750 atm. These two variables are related to each other through Henry's law.
C = k × P
K = C/P
K = (2.45 g/L)/0.750 atm = 3.27 g/L.atm
Step 2: Calculate the pressure required to produce an aqueous solution containing 6.25 g/L of this gas at constant temperature.
We have C = 6.25 g/L and k = 3.27 g/L.atm. The required pressure is:
C = k × P
P = C/k
P = (6.25 g/L)/(3.27 g/L.atm) = 1.91 atm
A solution at 25 degrees Celsius has a pH of 4.48. What is the pOH of this solution?
0.978
3.17
9.52
10.51
Answers
Answer:
The answer is C = 9.52
Explanation:
I took the test
The pOH of a solution that has a pH of 4.48 at 25°C is 9.52. Details about pH can be found below.
What is pH?pH refers to the power of hydrogen in a substance while the pOH refers to the power of hydroxyll ions. The pH of a solution ranges from 0 - 14 indicating the strength of acidity and alkalinity.
The relationship between the pH and pOH in a solution is given as follows:
pH + pOH = 14
According to this question, a solution at 25 degrees celsius has a pH of 4.48. The pOH of the solution is calculated as follows:
4.48 + pOH = 14
pOH = 14 - 4.48
pOH = 9.52
Therefore, the pOH of a solution that has a pH of 4.48 at 25°C is 9.52.
Learn more about pH at: https://brainly.com/question/15289741
Why when you get out of the pool, the cement pavement is hot compared to the pool water?
Answers
Answer: Water is a much better conductor of heat than air. So when you are surrounded by water, energy can flow from your body (conduction of heat) much more quickly than it would if you were surrounded by air at the same temperature.
If you have 1.4 grams of silver (Ag), how many moles of silver do you have?
Answers
Answer: There are 0.0129 moles of silver present in 1.4 grams of silver (Ag).
Explanation:
Given: Mass of silver = 1.4 g
Number of moles is the mass of substance divided by its molar mass.
As molar mass of silver is 107.86 g/mol. Therefore, moles of silver are calculated as follows.
[tex]No. of moles = \frac{mass}{molar mass}\\= \frac{1.4 g}{107.86 g/mol}\\= 0.0129 mol[/tex]
Thus, we can conclude that there are 0.0129 moles of silver present in 1.4 grams of silver (Ag).
Jenny has a gold bar in a piece of gold foil both objects are made of 100% pure gold watch and closing can guinea make about the two objects
Answers
Answer:
What is the question your asking?
Explanation:
All atoms of the same element have the same properties.
In order to use the Ideal Gas Constant of 0.0821, what units must be used for volume, pressure, amount, and temperature?
Answers
Answer:
The value becomes 0.0821 L-atm/K⁻¹-mol⁻¹.
Explanation:
The value of gas constant is 0.0821.
We know that, the ideal gas law is as follows :
[tex]PV=nRT\\\\R=\dfrac{PV}{nT}[/tex]
Where
P is pressure
V is volume
T is temperature
In order to use the Ideal Gas Constant of 0.0821, the units are follows :
Volume = Litre (L)
Pressure = atm
Amount = mol⁻¹
Temperature = K⁻¹
So, the value becomes 0.0821 L-atm/K⁻¹-mol⁻¹.
Which statement is true?
Answers
Answer:
A I think
Explanation:
Which of the following solutions is the most dilute.
a. 0.32 M
b. 1.5 M
c. 2.5 mol / L
d. 0.23 mol / L
e. 0.23 mol / L
Answers
Answer:
d and e
Explanation:
We have 5 solutions with different molar concentrations, that is, the quotient between the number of moles of solute and the liters of solution. This can be expressed as mol/L or M. The most dilute would be the one having the less number of moles of solute per liters of solution, that is, solution d or e, which have the same concentration. If we order them from the most diluted to the most concentrated, we get:
d = e < a < b < c
Lab 6: Basic Chemistry Thermodynamics: Solve the challenge of storing renewable energy
Answers
Answer:
Expensive and low storing capacity.
Explanation:
The storing of renewable energy is a big challenge for us because this renewable energy can't be stored for a long due to expensive and low storing capacity. There are two challenges about the storing of renewable energy is that the battery are very expensive and can store very less amount of energy which makes unable to store the renewable energy. The scientists should make a storing device which is not too much expensive and has more storage capacity.
Which of the following particles have the same mass. Proton, Neutron, Electron, None
Answers
Answer: proton and neutron
Explanation:
They both have the mass of 1
What type of bond will be formed between Ar and Cu
Answers
1.6 x 1023 atoms of Be, how many moles of Be are there?
Answers
Answer:
1 mole.
Explanation:
1.6x10^23 = NA = 1 mole of any substance.
The candle is lit and dilute ethanoic acid is poured down the inside of the beaker. As the acid reacts with the baking soda, bubbles of CO2 gas form. After a few seconds
the air in the beaker is replaced by 0.20 liter of CO2 gas, causing candle flame to go out. The density of CO2 gas is 1.8 grams per liter at room temperature.
Choose the correct structural formula for the acid that was poured into the beaker.
Answers
Answer:
It’s answer #2
Explanation:
I only know this because i just got it wrong
The correct structural formula for the acid that is poured into the beaker is CH₃COOH.
What is acetic acid?Acetic acid is also called ethanoic acid. It is form as a byproduct of fermentation. It forms vinegar. Vinegar is 4 to 6% acetic acid with water.
It is widely used in homes as cleaning agent, in cooking, washing etc.
Thus, the correct option is 2, CH₃COOH.
Learn more about acetic acid
https://brainly.com/question/15202177
#SPJ2
Convert 27.7 kilometers to centimeters.
Answers
Answer:
The correct answer is - 2770000 cm.
Explanation:
1 kilometer = 1000 meter
1 meter = 100 centimeter
1 kilometer = 100*1000 cm
1 km = 100000 cm.
then,
27.7 kilometers = 2.77 × 10^6 centimeters
So, 27.7 kilometers = 27.7 × 100000
= 2.77 × 106 or 2770000 centimeters.
PLEASE HELP!!! CHEMISTRY
Which one would require more energy to overcome because it is the strongest? Hydrogen bonding or van der waals?
Answers
How many atoms are in 1.20 moles of copper?
Answers
Answer:
7.23x10^23 atoms
Explanation:
1 mole = 6.023x10^23
1.20 mole = 6.023x10^23 x 1.2
= 7.23x10^23 atoms
The atoms present in 1.20 moles of copper is 7.23 × [tex]10^{23}[/tex] atoms.
What is atom?A chemical element is made up of atoms, which are the basic components of ordinary matter.
Calculation of atoms
Given data:
n = 1.20 moles.
It is known that.
1 mole = 6.023 × [tex]10^{23}[/tex]
So, 1.20 moles = 6.023 × [tex]10^{23}[/tex] × 1.2 = 7.23 × [tex]10^{23}[/tex] atoms.
Therefore, number of atoms in copper will be 7.23 × [tex]10^{23}[/tex] atoms.
To know more about atom click here.
https://brainly.com/question/1566330
#SPJ2
A student is trying to develop questions to help identify an unknown biome. Which of the following questions would provide helpful information to identify an unknown biome? Select ALL that apply. A. Is sunlight an abiotic factor in this biome? B. What is the average temperature of this biome? C. How much precipitation does this biome receive each year? D. What adaptations do plants and animals have to survive in this biome? E. Is the biome located near an ocean? F. Do plants and animals live in this biome? G. Does carbon cycle in this biome?
Answers
Answer:
I think the answer is C. D. And F.
Explanation:
I’m sorry if I’m wrong :(
Answer:
I think the answer is BCD and maybe F
Explanation:
Match the solutions to the descriptions of the freezing points.
a. One mole of the ionic compound Na3PO4 dissolved in 1000 g H2O
b. One mole of the ionic compound CuSO4 dissolved in 1000 g H2O
c. One mole of the nonelectrolyte C6H12O6 dissolved in 1000 g H2O
1. Highest freezing point
2. Intermediate freezing point
3. Lowest freezing point
Answers
Answer:
1c
2b
3a
Explanation:
Step 1: Write the general expression to calculate the freezing point depression
The freezing point depression is a colligative property, that can be calculated using the following expression.
ΔT = i × Kc × b
where,
i: van 't Hoff factor (number of ion particles per formula unit)
Kc: cryoscopic constant (Kc for water: 1.86 °C.Kg/mol)
b: molality (moles of solute per kilogram of solvent)
All the solution have the same Kc and the same b (1 mol/1 kg = 1 m), so ΔTf variation will depend on i
Step 2: Calculate the freezing point of the Na₃PO₄ solution
Na₃PO₄ has 4 ions (3 Na⁺ and 1 PO₄³⁻), so i = 4.
ΔT = i × Kc × b = 4 × 1.86 °C.Kg/mol × 1 mol/kg = 7.44 °C
T = 0°C - 7.44 °C = -7.44 °C
Step 3: Calculate the freezing point of the CuSO₄ solution
CuSO₄ has 2 ions (1 Cu²⁺ and 1 SO₄²⁻), so i = 2.
ΔT = i × Kc × b = 2 × 1.86 °C.Kg/mol × 1 mol/kg = 3.72 °C
T = 0°C - 3.72 °C = -3.72 °C
Step 4: Calculate the freezing point of the C₆H₁₂O₆ solution
C₆H₁₂O₆ is a nonelectrolyte (it doesn't ionize), so i = 1
ΔT = i × Kc × b = 1 × 1.86 °C.Kg/mol × 1 mol/kg = 1.86 °C
T = 0°C - 3.72 °C = -1.86 °C
Identify whether each compound is a straight chain, branched chain, or cycloalkane.
ОН
сн.
CH,
ж
E
НО шин-
-- ОН
нс
НО
"ОН
Answers
Answer:
Branched chain
Cycloalkane
Straight chain
Explanation:
What is the new concentration? L
M NaCl
Answers
Answer:
Explanation:
\we must convert the mass of NaCl in grams into moles. We do this by dividing by the molecular weight of NaCl (58.4 g/mole). Then, we divide the number of moles by the total solution volume to get concentration. The NaCl solution is a 0.1 M solution.
Answer:
0.125
Explanation:
boom
Calculate the number of molecules present in .20 moles of H2O
Answers
Answer:
Number of moles =20
Number of molecules present = 20 multipled by avogadro's number.
When does phenolphthalein turn pink?
in the presence of a base
in the presence of an acid
when it is in a neutral solution
when it is reacting with a metal
Answers
Answer:
A. In the Presense of a base
Explanation:
Just took the test from Edgunuity
Phenolphthalein turns pink in color in the presence of a base. Therefore, option A is correct.
What is phenolphthalein and its use?Phenolphthalein is a chemical compound that is often used as an acid-base indicator. It is colorless in acidic solutions and pink or red in basic solutions.
Phenolphthalein is often used in titrations to determine the endpoint of a reaction between an acid and a base. It can also be used as a laxative or in forensic investigations to test for the presence of blood.
Phenolphthalein has a chemical formula of C₂₀H₁₄O₄ and is synthesized by reacting phthalic anhydride with phenol in the presence of a strong acid catalyst. It is also used as a pH indicator. phenolphthalein has been found to have potential health benefits, such as reducing the risk of cancer, improving digestion, and reducing inflammation. Therefore, option A is correct.
Learn more about phenolphthalein, here:
https://brainly.com/question/15211751
#SPJ5
Que uso le darías a la vida diaria escribe en español plis
Answers
Explanation:
Facilitar el movimiento y desplazamiento de objetos pesados reduciendo el rose del objeto contra el suelo, Obtener un movimiento circular por efecto de la fuerza de un liquido, en el caso de contadores, molinos, centrales hidroeléctricas y turbinas.
Transmitir el movimiento de un eje a otro como en el caso de las lavadoras, bicicletas y neveras. Reducir drásticamente el esfuerzo necesario para elevar y mover objetos, como en casos de pozos de agua y ascensores Transformar movimientos giratorios en otros movimientos o viceversa. ,
Which is/are used in nuclear reactors to absorb neutrons?
A. heavy nuclei
B. critical mass
C. chain reactions
D. control rods
Answers
Answer:
A. heavy nuclei
Explanation:
They absorb neuyrons in order to hain stability.
By absorbing neutrons, a control rod prevents the neutrons from causing further fissions.
Why is medical technology good for society?
O A. It creates more jobs to do by hand,
B. It makes bilingspitals bigger.
C. It gives patients more illnesses.
D. It helps doctors treat diseases.
Answers
The ionization potential of Be atom is more than expected because it has -
(a) half filled valence p orbitals
(b) fully filled valence s orbitals
(c) both a and b
Answers
Answer:
(b) fully filled valence s orbitals
Explanation:
Electron configuration of Be: 1s22s2
2s2 is fully filled